Labrida AS enters agreement with Straumann Group
Norwegian BioTec start-up Labrida AS enters extensive agreement with Swiss based Straumann Group, a global leader in dental care.
- Labrida AS and Straumann Group agreed on an exclusive distribution agreement in key markets for Labrida BioClean™, a medical device class 2a for the maintenance of dental implant health.
- Straumann Group has further strengthened its collaboration with Labrida by acquiring a minority stake in Labrida.
The agreement with Straumann is a milestone for Labrida. Being a biotec start-up, with a product we believe in, we have an ambition to make our innovative solution accessible for dentists globally. With Straumann we found a powerful partner with a global distribution reach which is key. We are therefore proud to announce such an extensive agreement with one of the true benchmarks of the industry
BaghLabrida AS and Straumann Group have entered into an agreement providing Straumann exclusive distribution rights in key markets for Labrida BioClean™ – a professional brush for managing long-term maintenance of implants. In 2021, Labrida and Straumann wish to expand the distribution agreement to more countries, mainly in Asia Pacific and emerging markets. Furthermore, Straumann Group has acquired a minority stake in Labrida and obtained option rights to increase its shareholding in the future. This allows Labrida to further invest into research and development as well as to scale-up production.
“The agreement with Straumann is a milestone for Labrida. Being a biotec start-up, with a product we believe in, we have an ambition to make our innovative solution accessible for dentists globally. With Straumann we found a powerful partner with a global distribution reach which is key. We are therefore proud to announce such an extensive agreement with one of the true benchmarks of the industry”, says Tore Heldrup Rasmussen, Chairman of the Board of Labrida.
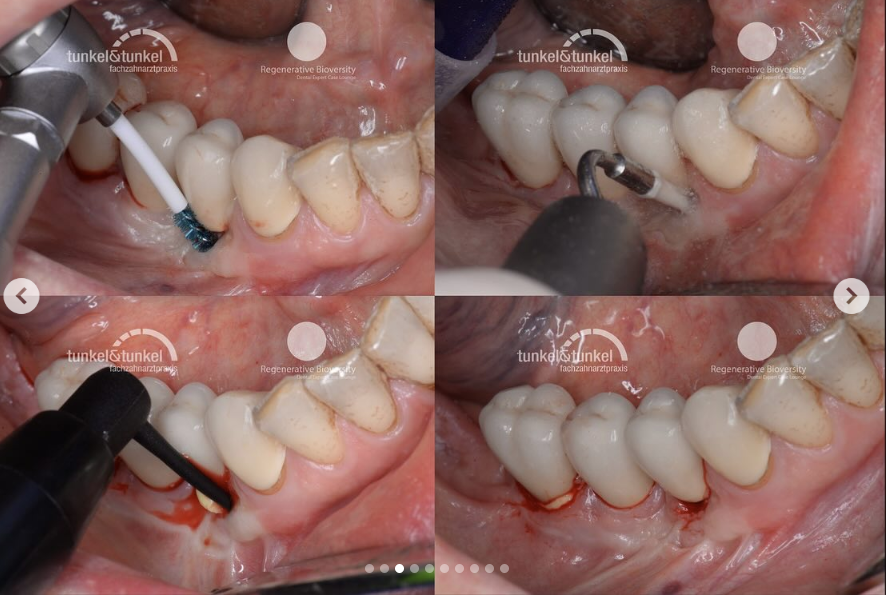
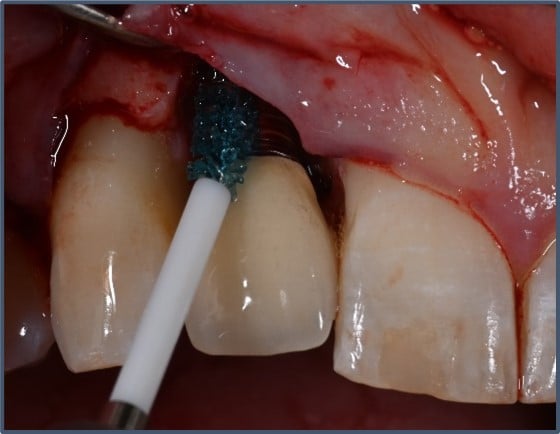
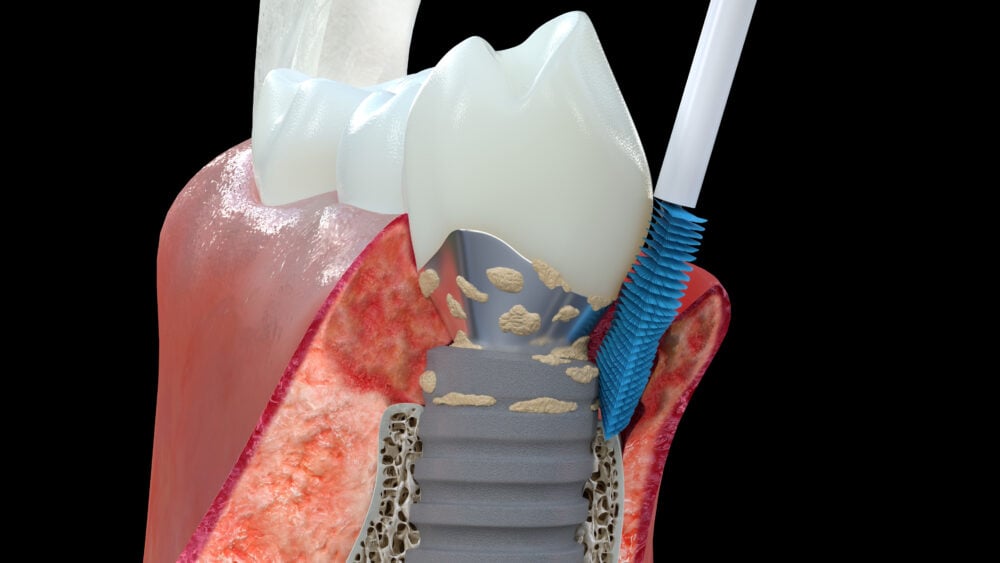
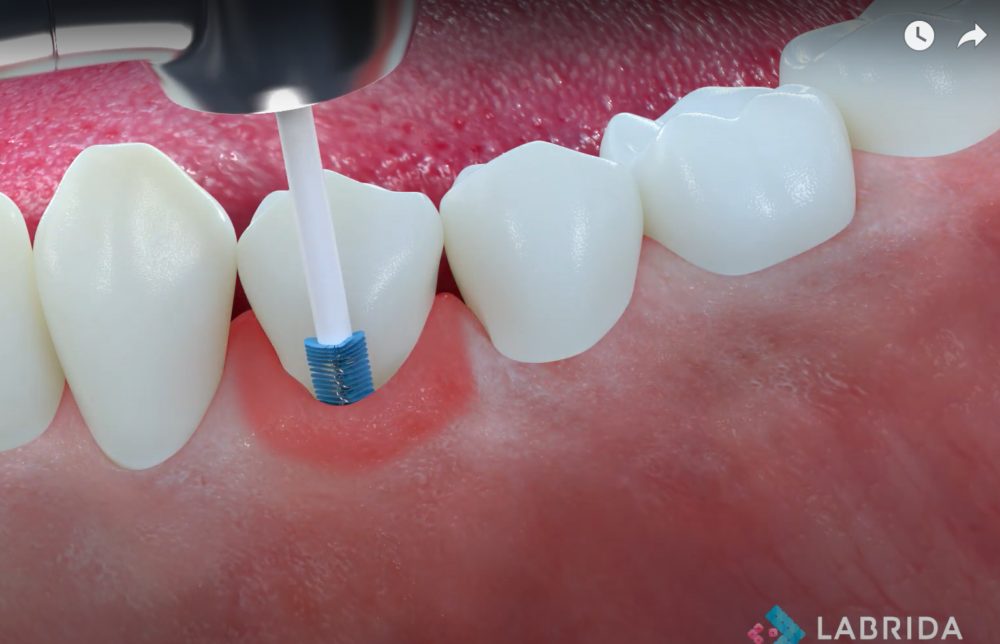
Labrida has developed, registered, and is marketing the Labrida BioClean™ brush for professional use, helping to significantly reduce inflammation, documented both in cases of peri-implant mucositis and mild peri-implantitis. The brush is made of the marine based substance chitosan, a biomaterial used for implant supportive care. A study performed by SINTEF, a Norwegian research institution, showed that Labrida BioClean® further helps to reduce the release of aerosols compared to traditional treatments, providing an additional option for dentist to meet newly imposed measurements for dental clinics due to COVID-19.
“The new partnership is truly a win-win for both companies. With the innovative professional brush for managing implant health, we will be able to offer dentists worldwide an additional solution for the maintenance of implants. Implant health is important to avoid inflammations or other complications in the long-term. The collaboration with Labrida allows us to extend our portfolio in implant health and to offer additional solutions for managing peri implant diseases.”, says Holger Haderer, Head of Marketing & Education at Straumann Group.
Labrida BioClean™ is an alternative to existing treatments for the maintenance of dental implants. The implant health market is estimated to be about 70 million individuals worldwide, each patient typically needing 2-4 treatments per year. Labrida BioClean™ is also approved for periodontitis, a gums inflammation. This market is estimated to about 450 million individuals worldwide, and as such considered to be the world’s 6th largest disease by incidence.
“We were hit hard by covid-19, as dental practises all around the globe were shut down overnight. Nonetheless, we have used this downtime productively and with the tireless support of our entire team, Innovation Norway, and of course the team at Straumann, we are now heading into 2021 with high hopes and expectations for strong commercial progress”, says Arvid Lindberg CEO of Labrida.
The agreement was mutually signed on December 18th 2020, and product launches throughout a vast number of European countries has already been either initiated or planned for 2021.